Medical Desiccants

Desiccant for Medical Industry
Industry Challenges
Hydrolytic Degradation of APIs
- Active Pharmaceutical Ingredients (APIs) are highly susceptible to moisture. Even trace humidity can trigger chemical reactions that reduce potency, alter dissolution rates, and render life-saving medications ineffective before they reach the patient.
Sterility Barrier Integrity
- Maintaining a sterile environment is critical. Excess moisture within sealed medical packaging can compromise Tyvek® seals and adhesive bonds, creating pathways for bacteria and contaminants to enter the sterile field.
Condensation in Diagnostic Kits
- Diagnostic reagents and test strips are humidity-sensitive. Temperature fluctuations during shipping can cause internal condensation, leading to false positives/negatives, clotting in microfluidics, or the premature activation of chemical reagents.
Corrosion of Surgical Instruments
- High-value surgical tools and electronic medical devices suffer from electrochemical corrosion when exposed to humid environments. Rust or oxidation can damage fine-tuned mechanisms, posing significant surgical risks.
Why Choose Our Desiccants
Pharmaceutical Compliance
Manufactured in accordance with ISO 13485 standards, ensuring traceability and safety for direct contact with pharmaceutical packaging.
Customizable Formats
Available in Tyvek® bags, canisters, or adhesive pads to fit seamlessly into automated high-speed blister and vial filling lines.
Contaminant-Free
Our pouches feature low-dust formulations and are free from heavy metals and volatile organic compounds (VOCs) to protect sensitive reagents.
Predictable Kinetics
Engineered with precise adsorption capacities (clay, silica gel, molecular sieve) to maintain specific Relative Humidity (RH) targets between 10% and 30%.
Usage
Pharmaceutical Packaging (Vials, Blister Packs)
Overview
Protecting solid dosage forms such as tablets and capsules, as well as moisture-sensitive powders, inside sealed vials and blister cavities by maintaining precise low-humidity conditions throughout the product's shelf life.
Key Advantages
- Extended Shelf Life: Maintains low RH to prevent API hydrolysis and preserve drug efficacy for the labeled shelf life.
- High-Speed Integration: Pre-formed canisters and continuous coil pouches allow for automatic insertion without slowing down blister line speeds.
- Child-Resistant Compatibility: Designed to fit within standard blister cavity depths without compromising child-resistant closure mechanisms.
- Material Safety: Utilizes non-toxic, pharma-grade desiccants that meet USP standards for packaging systems.
Medical Device & Instrument Protection
Overview
Safeguarding reusable surgical instruments and single-use electronic medical devices from moisture-related damage during sterilization processes, storage periods, and transportation across various environmental conditions.
Key Advantages
- Corrosion Prevention: Adsorbs residual moisture post-autoclave sterilization, preventing rust on hinges, blades, and fine surgical tips.
- Electronic Integrity: Protects internal circuitry and battery contacts in powered surgical drills and monitoring devices from moisture-induced short circuits.
- Tyvek® Compatibility: Pouches bonded to Tyvek® allow for ethylene oxide (EtO) sterilization gas penetration while blocking liquid and bacterial ingress post-sterilization.
- Secure Placement: Adhesive-backed desiccants ensure the drying agent stays affixed inside the instrument tray during transport and handling.
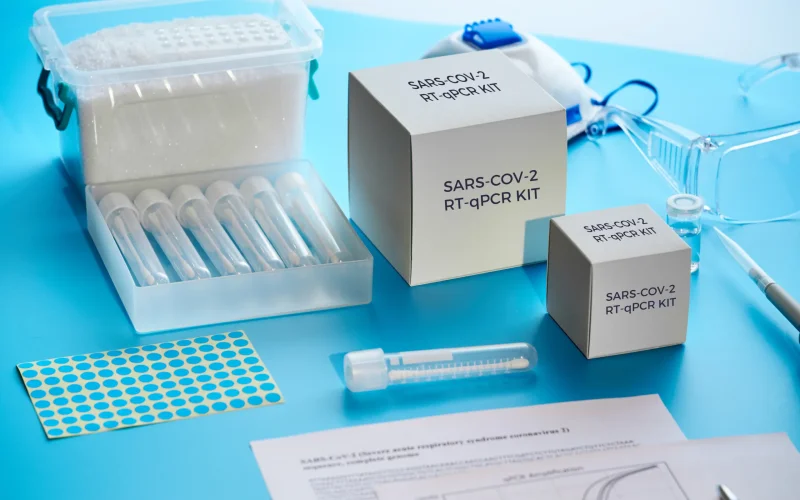
Diagnostic Reagent & Test Kit Preservation
Overview
Ensuring accuracy and reliability in lateral flow assays, microfluidic cartridges, and dry reagent storage by preventing humidity-induced degradation that could compromise diagnostic performance and results.
Key Advantages
- Reagent Stability: Prevents deliquescence (caking) of lyophilized reagents, ensuring consistent flow rates across test strips.
- False-Negative Reduction: Maintains ultra-dry conditions to prevent antibody denaturation in rapid diagnostic tests (RDTs).
- Low-Temperature Shipping: Stabilizes the micro-climate during cold-chain breaks, protecting enzyme-based reagents from humidity fluctuations in transit.
- Non-Interference: Formulated to be free from particulates that could obstruct micro-channels or contaminate optical reading surfaces.
Implantables & Sterile Barrier Systems
Overview
Supporting the integrity of sterile barrier systems for orthopedic implants, surgical sutures, and tissue grafts by controlling moisture levels that could compromise seal strength and sterility assurance.

Key Advantages
- Seal Integrity: Prevents "pop-open" failures of sterile pouches by controlling moisture expansion that weakens heat-sealed boundaries.
- Cleanroom Compatibility: Produced and packaged in ISO Class 7 cleanroom environments to match the hygiene standards of implant manufacturing.
- Biocompatibility: Utilizes materials suitable for indirect contact with human tissue (ISO 10993-1), ensuring no cytotoxic leachables.
- Rigorous Validation: Each batch undergoes stringent adsorption capacity testing to provide validated documentation for regulatory submissions (510k, CE Mark).
